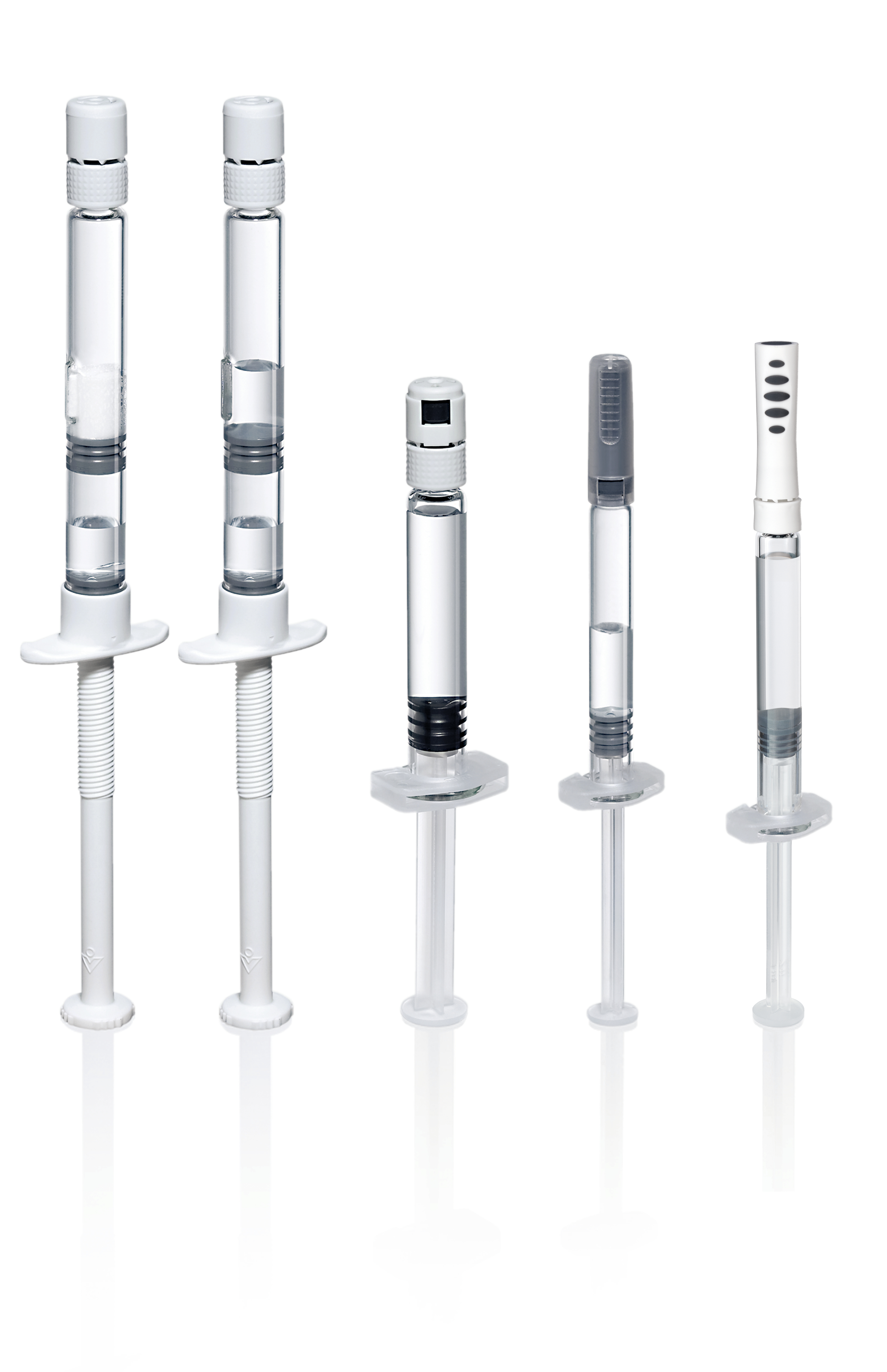
Injection systems add value at clinical and commercial stages. Choosing the right type of packaging is crucial
Increased competition among leading brands of injectable drugs such as novel therapeutics and biosimilars is driving the need for product differentiation. One way to achieve market advantage is to move from a classical vial presentation to a prefilled syringe. Syringes offer innovative delivery options, help achieve market advantages at launch, and act as a valuable life cycle management strategy.
In this webinar we will examine the advantages of prefilled syringes, including the various differentiators and most commonly used syringe platforms, both pre-sterilized and bulk. Factors to be considered in making the right choice of prefilled syringe formats will be revealed. Customization in syringe packaging materials and production steps, along with techniques for coping with more challenging compounds, e.g., sensitivity to silicone or reduced particle load, will be reviewed. Innovations in prefilled syringes that help meet today's market challenges will be highlighted.
By attending this webinar the participant will:
- Gain an understanding about the two prefilled syringe platform technologies on the market: pre-sterilized syringes and bulk syringes
- Learn how prefilled syringes can offer distinct advantages in product differentiation and life cycle management strategies
- Learn how to choose the syringe system for your compound and how prefilled injection devices, which have been designed with safety and simplicity in mind, might best be incorporated into a drug development program.
This webinar will be presented live twice at:
- 8AM London/3PM Singapore/4PM Tokyo
- 3PM London/10AM New York/9AM Chicago
Please register for the most suitable time.
Presented by

Kevin Wrigley,
Product and Service Manager, Vetter Pharma International GmbH
Kevin Joseph Wrigley earned his MSc in International Business from the University of Ulster, UK in 2009. At Vetter, he is responsible for managing the commercial manufacturing business as it pertains to the product lifecycle and portfolio management perspective.
Kevin began his career in the medical device and pharmaceutical industries in management positions at Smith & Nephew, and MorphoSys. He joined Vetter International in 2013 as Product & Service Manager.

Joerg Zimmermann,
Vice President Vetter Development Service, Vetter Pharma-Fertigung GmbH & Co. KG
Joerg Zimmermann studied pharmacy in Freiburg, Germany and Cardiff, Wales, and joined Vetter in 1994 as assistant head of production. His first responsibilities were process development and new product introductions with a focus on lyophilization process development.
For more than 10 years, Joerg was responsible for the Vetter Site in Langenargen as Director of production, a site with 5 filling lines for aseptically prefilled injection systems. In September 2010, he moved into the role of Director Process Development and Implementation.
In 2014, he became Vice President of Vetter Development Service in Germany, covering all aspects of pharmaceutical process development and qualification, development of packaging components, analytical method transfers, and stability studies.